Abstract
The mass loading of silicon-graphite electrodes is often considered a secondary parameter when testing their electrochemical performance. However, if sacrificial additives are present in the electrolyte to improve electrochemical performance, the electrode loading becomes the determining factor in battery cycle life. The correlation between mass loading, electrolyte additives and binder type was investigated by analyzing the cyclic behavior of silicon graphite electrodes prepared with water-based binders with mass loading ranging from 3 to 9.5 mg cm -2 and used FEC electrolyte circulation additive while maintaining electrolyte quality constant. By keeping the slurry preparation steps constant for different adhesives, lower loads can be achieved and result in longer service life for some adhesives. In contrast, when the final load is held constant, the performance becomes independent of the adhesive used. Since such results may lead to a misunderstanding of the effect of electrode composition on cycling stability (and in our case to the superiority of one binder over another), we recommend that long-term electrochemical testing of silicon-graphite electrodes be Chemical performance data for comparison need to be always collected by using the same mass loading and constant electrolytes and additives.
Growing demand for lithium-ion batteries (LIBs) has prompted researchers around the world to develop simple but feasible ways to increase the gravimetric and volumetric energy of commercial batteries. One way to achieve this is to increase the specific charge of the negative electrode. The simplest method is to mix graphite (Q th = 372 mAh g -1 ) with a specific charge-enhancing component (i.e. an element or compound with a significantly higher specific charge than graphite), such as silicon (Q th = 372 mAh g -1 1 ). = 3572 mAh g -1 ). Although on the one hand, the resulting composite electrode offers a higher specific charge, on the other hand, the silicon-based electrode has poor cycling stability due to volume changes during alloying of silicon with lithium. Volume changes can cause cracks in the solid electrolyte interphase (SEI) layer, resulting in continued depletion of electrolyte at the electrode surface and progressive loss of electronic contact between the electroactive material particles and the conductive carbon.
Cycling stability can be improved by preparing electrodes using preferably water-processable binders, such as polyacrylic acid (PAA), sodium carboxymethylcellulose (Na-CMC), and sodium alginate, as they are able to partially buffer the Si volume due to cross-linking. Swelling occurs due to the formation of interconnected networks and strong interaction with silicon particles.Furthermore, the addition of additives to the electrolyte has been shown to improve the cycle life of alloy-based electrodes. Additives such as fluoroethylene carbonate (FEC) can form SEIs that are thinner, more uniform, and more mechanically stable than those formed in ordinary carbonate electrolytes.
The long-term cycling stability of silicon-graphite electrodes in the presence of additives indirectly depends on another important parameter: the electrode mass loading. Since additives are often sacrificial and are consumed during cycling, the lifetime of cells containing silicon electrodes has been shown to be linearly related to the ratio between additive and active material loading.This means that using the same amount of electrolyte, a larger amount of additive or a lower mass loading will result in a higher number of cycles. Furthermore, it should be taken into account that the amount of electrolyte in research cells is much higher than in commercial cells, so the same concentration of additives in the electrolyte will result in significantly different absolute amounts of additives available for interface protection and the amount of active additives. Material loading is more important. However, electrode loading is an often overlooked parameter in many scientific publications. Literature reports showing unloaded or, more commonly, Si or Si-graphite containing electrodes are reported with a very wide range of loading values (1 to 10 mg cm -2 or 0.1 –2.5 mg considering the total amount of electroactive material cm -2 only considers the amount of Si). There are two ways to perform comparable experiments: either keep the procedure unchanged or keep the final test setup unchanged, in this case either the slurry formulation or the mass loading of the final electrode. Another approach is to adjust the amount of electrolyte and additives according to the load, in which case the reliability of the experiment is less stable.
Therefore, the purpose of our study was to demonstrate the interrelationship between electrolyte additives, electrode loading, and binder type, and how these parameters affect the cycling stability of silicon graphite electrodes. Furthermore, we would like to make the battery community aware of the need for standard performance comparisons of silicon-graphite electrodes, which must take into account the amount of additives used and the loading of the electrodes.
experimental
Material
The following materials were used for electrode preparation: Si nanoparticles (30–50 nm, 98%, amorphous and nanostructured materials), C-NERGY KS6L graphite (d 90 = 6.5 μm , S BET = 18 m 2 g −1 , Imerys graphite and carbon), C-NERGY SuperC45 conductive carbon (SC45, average particle size = 37 nm, S BET = 45 m 2 g −1 36 ), polyacrylic acid binder mixture (PAA, 25 wt% solution in water, avg. Binder mixture of 1:1 molecular weight 240,000 Alfa Aesar) and carboxymethyl cellulose (CMC, Alfa Aesar), 1:1 CMC and styrene-butadiene rubber (SBR, MTI Corporation), guar gum mixture (Sigma-Aldrich), sodium alginate binder (Sigma-Aldrich).
Electrode preparation
The weight ratio of the active and inactive components of the electrode (graphite: Si: SuperC45 carbon: binder) is 90:5:1:4.
In standard formulations, the adhesive is first dissolved in a solvent (a mixture of water:ethanol 70:30 wt% in the case of PAA:CMC, CMC:SBR and sodium alginate, and in the case of PAA:CMC, CMC: SBR and sodium alginate, then use only water) guar gum), then stir the SC45 conductive additive and Si nanoparticles into the binder solution. Then, add graphite powder to the mixture and stir the slurry until you get a honey-like texture. Stirring was performed by a turbine stirrer (IKA Ultra-Turrax T25). After degassing on the drum mixer for 2 hours, the slurry was cast onto copper foil using the doctor blade technique. For all binders, the obtained electrode sheets were dried under vacuum at 80°C overnight, and for the PAA:CMC binder an additional 2 h were pretreated at 150°C, since at this temperature the binder mixture Cross-linking occurs. 7 Punch out a 13 mm diameter round electrode and re-dry under vacuum at 120 °C overnight to remove any remaining traces of water.
Medium and Coarse Grain---Special Graphite Block/Round
Electrochemical characterization
The electrodes were assembled into a button cell in an argon-filled glove box (<0.1 ppm H 2 O and <0.1 ppm O 2 ), with metallic lithium (≥99.9%, thickness 0.75 mm, Alfa Aesar) as the counter electrode, and glass fiber separators. plate. The following electrolytes were used in this study: 1 M LiPF 6 in ethylene carbonate (EC): dimethyl carbonate (DMC) (1:1) (LP30, BASF) + 2 wt. fluoroethylene carbonate (FEC) ) (BASF).
The ratio of FEC to electroactive material (EAM = graphite + Si) is calculated by dividing the molar amount of FEC in the electrolyte (expressed in μmol) by the mass of electroactive material in the electrode (in mg) respectively . Electrochemical measurements were performed at 25 °C using a battery cycler (Astrol Electronics AG, Switzerland) with the following cycling procedure: a first constant current (CC) cycle was performed at a low rate (20 mA g -1 ) to allow SEI formation, while undergoing further CC cycles for lithiation at 50 mA g and delithiation at 186 mA g . After each CC step, the cutoff potential (5 mV for lithiation; 1.5 V for delithiation) was maintained until the current dropped below 5 mA g ( constant voltage (CV) step). In this study, the electric potential is referred to Li + /Li, and the specific charge is expressed per mass of electroactive material (i.e., the sum of the masses of graphite and Si). To ensure reproducibility, cycle data from at least two nominally identical cells were considered for each given set of conditions. For clarity of presentation, only the specific charge obtained during delithiation is shown in the performance graphs. Table I summarizes the electrodes studied and the terminology used in this article .
Table I. Overview of the electrodes studied. The electrode composition is always graphite:Si:SC45:binder = 90:5:1:4. R and U represent "regular" and "unified" loads respectively.
| Name | Binder (4% total) | Average load ±0.5 (mgEAM cm −2 ) | Solid content (%) | Blade clearance ( μm ) | Average thickness ±2 ( μm ) | Average density ±0.02 (g/cm3) | Estimated porosity ±1(%) |
|---|---|---|---|---|---|---|---|
| PAA: CMC | 2 wt% PAA:2 wt% CMC | 9.8 | 31 | 250 | 165 | 0.62 | 72 |
| CMC:SBR_R | 2 wt% CMC:2 wt% SBR | 7.1 | 27 | 250 | 130 | 0.57 | 74 |
| GG_R | 4 wt% guar gum | 5.4 | 23 | 250 | 90 | 0.64 | 71 |
| alg_R | 4 wt% sodium alginate | 3.7 | number 17 | 250 | 70 | 0.55 | 75 |
| CMC:SBR_U | 2 wt% CMC: 2 wt% SBR | 9.3 | 27 | 400 | 155 | 0.63 | 71 |
| GG_U | 4 wt% guar gum | 9.7 | 23 | 450 | 160 | 0.63 | 71 |
| Alg_U | 4 wt% sodium alginate | 9.2 | 23 | Chapter 470 | 165 | 0.58 | 73 |
Results and discussion
This study cycles Si-graphite electrodes with KS6L graphite, Si nanoparticles, PAA:CMC binder, and approximately 9.5 mg cm loading in LP30 + 2 wt% FEC electrolyte following the CCCV protocol. This choice resulted from the extensive research carried out by our team over the past few years on this type of electrode; specifically, optimizing the type of silicon , cycling scheme The composition of the electrode (i.e. silicon amount and graphite type) maximizes electrochemical performance. To further improve the cycling stability, electrodes with different water-processable binders were produced (Table I ), tested, and compared with the baseline.
The potential distribution of the first cycle is shown in the figure, while the electrochemical performance is shown in Figures 1a and 1b. 1b and 1c and are summarized in Table II .
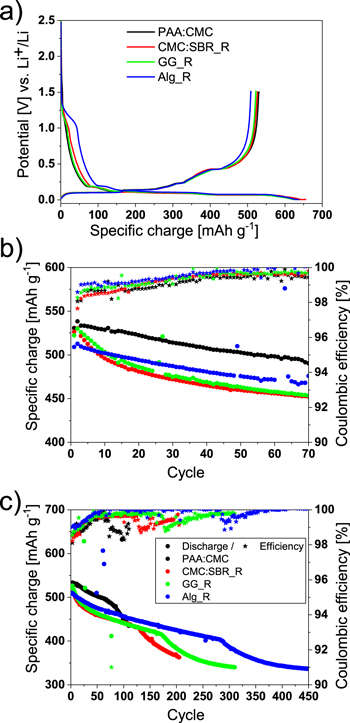
Figure 1. (a) first cycle potential distribution, (b) short-term and (c) long-term electrochemical performance (blue) of PAA:CMC (black), CMC:SBR_R (red), GG_R (green) and Alg_R. of).
Table 2. Electrochemical data of the studied electrodes.
| Specific charge, mAh g −1 (capacity retention rate, %) | |||||
|---|---|---|---|---|---|
| Name | Second delithiation | 70 | 200 | 300 | 450 |
| PAA:CMC_R | Chapter 538 | 490 (91) | 0 | 0 | 0 |
| CMC:SBR_R | Chapter 530 | 453 (85) | 366 (69) | 0 | 0 |
| GG_R | Chapter 530 | 453 (85) | 383 (72) | 342 (65) | 0 |
| alg_R | Chapter 512 | 468 (91) | 425 (83) | 387 (75) | 336 (66) |
| CMC:SBR_U | Chapter 526 | 463 (88) | 0 | ||
| GG_U | Chapter 530 | 456 (86) | 0 | ||
| Alg_U | Chapter 533 | 482 (90) | 0 | ||
For all investigated electrodes with different types of binder, the potential profiles show the characteristic reduction of FEC at around 1.2 V during the first lithiation. As the lithiation proceeds, the plateaus, related to the alloying of Li with Si, and the intercalation of Li into graphite, are visible below 0.3 V. During the 1st delithiation, the three plateaus associated with graphite are visible up to 0.25 V and then, at 0.45 V, the plateau related to the delithiation of crystalline Li15Si4 phase is clearly distinguishable. Interestingly, the potential profile in the proximity of FEC reduction potential differs slightly depending on the binder composition. The differences can be better seen in the differential capacity plots (Fig. S1). The noticeable reduction of FEC starts earlier at a higher potential for CMC:SBR_R and Alg_R electrodes, specifically at around 1.35–1.4 V; while it starts around 1.15–1.2 V for PAA:CMC_R and GG_R electrodes. Furthermore, electrodes with Na-Alginate binder show a much more pronounced FEC reduction, hence a higher irreversible specific charge associated to SEI formation in comparison with the other binders. The 1st cycle columbic efficiency is around 80%–82% for PAA: CMC, CMC:SBR and GG, while below 80% for Na-alginate. The higher reactivity of FEC towards alginate-based electrode might be explained by poorer alginate binder coverage of Si particles, and therefore higher electroactive area for FEC reduction, while the higher potential for FEC decomposition onset is most likely because of the higher decomposed FEC amount, which makes detection already at higher potentials possible. The first signs of FEC reduction have been shown as early as at 1.5 V.41
PAA:CMC electrodes provide a specific charge of around 540 mAh g−1 at the end of the 2nd delithiation, which is the highest among the electrode with studied binders and is also very close to the theoretical one of 540.8 mAh g−1. Alg_R shows the lowest specific charge on the first discharge (512 mAh g−1) indicating that roughly 15% of the Si nanoparticles are not participating to the reaction already from the initial cycles, which points to the poorer binder distribution and Si domains, inaccessible to either electrolyte or electrons. CMC:SBR_R and GG_R show a specific charge of around 530 mAh g−1.
The cycling behavior of the electrodes was analyzed in the short (70 cycles, Fig.1b) and long-term (450 cycles, Fig.1c). During the first 70 cycles, both PAA:CMC and Alg_R electrodes fade with a nearly constant slope reaching a specific charge retention of 91%. CMC:SBR_R and GG_R electrodes instead show a rapid specific charge drop for the first 10 cycles, and successively the fading continues with a constant slope in the same way as electrodes with PAA:CMC and Na–Alginate binder. We hypothesize that the rapid fading might be the result of a different kind of interaction between Si particles and these two binders, as they lack the carboxyl groups, which have been shown to have a positive effect on cycling stability.42Indeed, guar gum and SBR do not have carboxyl groups and absence or reduction of carboxylic groups might be the reason for adversely affected initial cycling stability. The specific charge retention after 70 cycles is around 85% for electrodes with CMC:SBR and GG binder.
On the long term, all electrodes show a very similar behavior: a constant fading for a certain number of cycles, then a rapid fading known to be caused by the depletion of FEC, which is accompanied by a decrease in columbic efficiency.14After the rapid fading, the specific charge converges to contribution of only graphite, showing that almost all of Si is by now inactive and presumably disconnected due to the surrounding SEI layer from the electronic conductive network. Ultimately, most of the cells completely fade either because of a short circuit caused by dendrite formation at the lithium counter electrode43or because the electrolyte consumption (Fig. S2).
Electrodes with Na-alginate binder seem to provide the best specific charge retention reaching 65% after 450 cycles, while electrodes with other binders are not even able to reach such a high cycle numbers. Even though at a first glance it seems that a straightforward conclusion is that Na–Alginate is the best binder for Si–graphite electrodes, we took it with caution due to difference in active materials loading, as from our previous studies, we have seen that experimental parameters can have a significant influence on the final performance of the cell.44
As a first attempt to clarify if the loading has effect on cycle life and capacity retention, while using the same amount of electrolyte, and thus additive, we have plotted in Fig.2a ratio between the FEC amount and the electrode loading vs the cycle number corresponding to the onset of rapid fading (i.e., the last cycle before the columbic efficiency significantly drops). The result showed that the two parameters (i.e., the FEC/EAM ratio and the onset of rapid fading) are linked via a linear correlation: the higher active mass loading, the shorter is the cell's lifetime, and that this correlation is independent of the type of the binder. A linear correlation was already reported for Si electrodes cycled with different amounts of an electrolyte additive14but our finding explain interrelation between electroactive materials loading, additive/electrolyte amount, and the type of binder. Based on this we can conclude that our results were the consequence of a clear and implicit trend: only the electrode loading and the electrolyte additive amount are decisive, while the electrochemical cycling results are independent of the binder used.
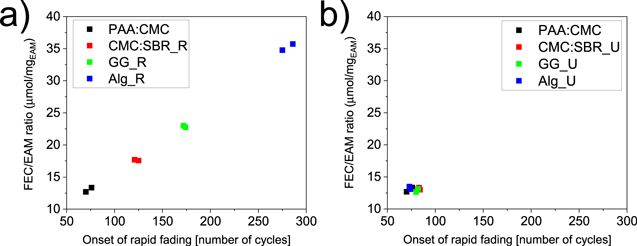
Figure 2. FEC/EAM ratio versus onset of rapid fading plots for a) regular electrodes and b) unified electrodes of PAA:CMC (black), CMC:SBR_R (red), GG_R (green) and Alg_R (blue).
Download figure:
Standard image High-resolution imageTo experimentally verify our conclusion, all electrodes, earlier prepared using our standard procedure with different binders, while having different loadings, now were prepared adjusting the electrode preparation procedure to give comparable loadings to our baseline one (around 9.5 mg cm−2). The potential profiles for the 1st cycle are shown in Fig.3a and the electrochemical performance in Fig.3b. The higher loading, obviously, does not change the potential profile of the electrodes, however a slight difference regarding FEC reduction is observed for Alg_U electrodes in comparison to lower-loading Alg_R electrodes. The higher loading shifts the signature of FEC reduction by ca. 0.1 V towards lower potentials (Fig. S3). The same effect was reported for Si electrodes prepared with only PAA or only CMC binders by increasing Si loading 2.5 times.45In our study, even though the Si amount was increased also 2.5 times, this effect was observed only with Na-Alginate. CMC:SBR_U and GG_U electrodes, show a specific charge after the 2nd delithiation of around 530 mAh g−1, very close to that one of the lower loading electrodes with the same types of binder. However, electrodes with Na-Alginate show an improvement in specific charge reaching 533 mAh g−1. The lower amount of solvent (i.e., higher solid content) necessary to obtain a higher loading resulted in a more homogeneous binder and carbon distribution in the electrode,46as well as a better connectivity between EAM and SC45 particles.47This leads to a higher amount of Si being exploited and resulted in a higher specific charge.
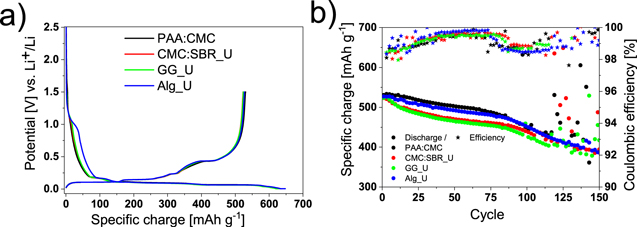
Figure 3. (a) potential profile for the 1st cycle and (b) electrochemical performance of PAA:CMC (black), CMC:SBR_U (red), GG_U (green) and Alg_U (blue).
Download figure:
Standard image High-resolution imageAs for the cycling stability, high loading electrodes mimic the behavior of lower-loading electrodes in the initial 70 cycles. Alg_U shows a constant fading over cycling, while CMC:SBR_U and GG_U show a rapid drop during the initial 10 cycles, and then the specific charge drops in a constant fashion.
The pronounced difference in specific charge retention between electrodes with different binders (as shown in Fig.1) is not observed when electrodes have all the same mass loading. High loading electrodes have nearly the same fading rates, independently of the type of the binder. The specific charge retention after 70 cycles is around 90%–91% for electrodes with PAA:CMC and Na-Alginate binders, and around 86%–88% for CMC:SBR and GG binders (TableII).
The complete consumption of FEC occurs at about the same number of cycles for all electrodes with ca. 9 mg cm−2 loading, i.e., after 70––80 cycles. The rapid fading after FEC consumption leads to a specific charge of around 380–390 mAh g−1 after ca. 150 cycles and ultimately to complete cell failure after ca. 200 cycles for all binders (Fig. S4). The relationship between FEC/EAM ratio and rapid fading is visualized in Fig.2a. It is now clear that, for Si-graphite electrodes, Na-Alginate is not a better performing binder than PAA:CMC but the additive amount and the loading are the two factors determining the cell lifetime.
Thus, it is obvious that the electrode loading plays an important role in the evaluation of electrochemical performance when electrolyte additives are used. A wrong interpretation of electrochemical data from electrodes with different loadings can actually disguise the importance of underlying experimental parameters. As an example, in Ref.48, the low capacity retention of the electrodes with Na-Alginate was assigned to the binder but the fading is instead related to the complete consumption of the additive at a specific loading. Moreover, as FEC is the state-of-the art electrolyte additive for Si-graphite electrodes in both research and industry, and more and more scientific publications report electrochemical performance with the use of this electrolyte additives, the care should be taken to unify the testing parameter. We believe that an objective comparison of long-term electrochemical performance between Si–graphite electrodes can only be done by using the same amount of electrolyte additive and comparative loadings.
Conclusions
The results of this study indicate that load plays a major role in determining battery cycle life in the presence of a constant amount of electrolyte additive. Electrodes with different mass loading and different types of binders faded after many cycles, linearly with the additive/loading ratio, and contrary to expectations, the cell cycle life did not depend on binder type. In this case, the difference in cycle life is small when the electrodes are used with different binder types but the active material loading values are very close, which leads to the conclusion that the FEC consumption per unit amount of active material is the same for each cycle , independent use of adhesive. This means revisiting past results where sacrificial additives were used to consider binder properties as performance enhancing factors, and experimental conditions, especially differences in electrode loading and electrolyte (additive) to active material ratio, should be carefully scrutinized. Our results also suggest that to test the effect of changes in electrode composition (e.g. binder in our case), the same active material loading should be the guiding parameter rather than the same electrode preparation conditions.
